
Mostbet букмекерская контора на реальные деньги
Mostbet — известная букмекерская контора, завоевавшая доверие тысяч игроков по всей стране. Ставки на спорт, разнообразные бонусы, удобный интерфейс сайта и мобильное приложение делают её популярной среди поклонников азартных игр и спортивных событий. Работая на рынке уже множество лет, Mostbet постоянно совершенствует свои услуги, чтобы предоставить своим клиентам высокое качество и безопасность.
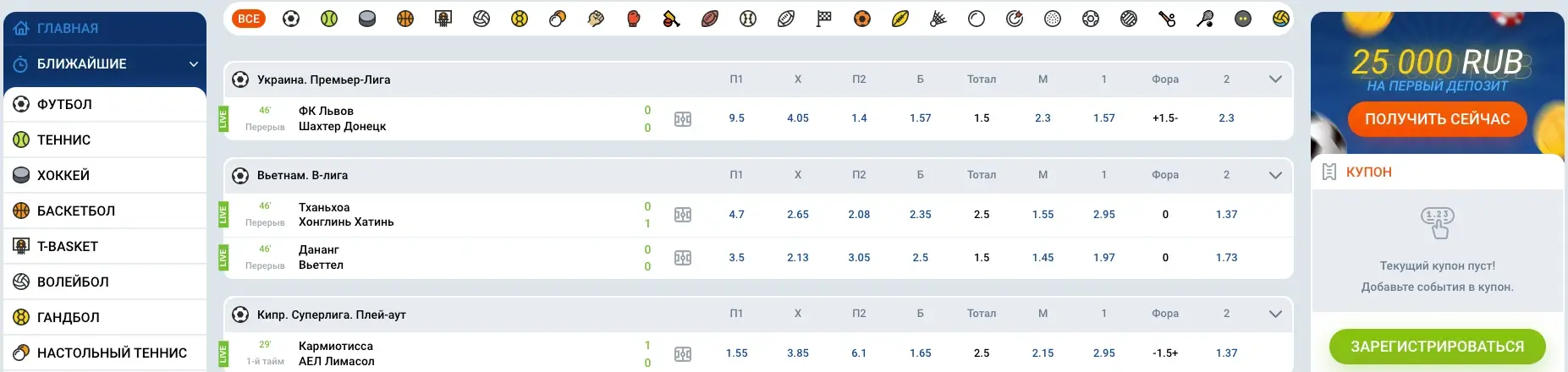
В Mostbet online вы найдете широкий ассортимент ставок на спортивные события: футбол, хоккей, баскетбол, теннис, биатлон, гонки и многое другое. Контора предлагает участникам выгодные коэффициенты и оперативное обновление линий. Благодаря простой регистрации и возможности делать ставки в режиме онлайн, пользователи могут наслаждаться азартом и острыми ощущениями от соревнований в любое удобное время.
| Год основания | 2009 |
| Лицензия | Curaçao eGaming |
| Тех. поддержка | Телефон, электронная почта, онлайн-чат |
| Мин. депозит | 100 руб. / 2 долл. США / 2 евро |
| Мин. вывод | 500 руб. / 10 долл. США / 10 евро |
| Валюты | RUB, USD, EUR и другие |
| Типы бонусов | Приветственный бонус, бонусы за депозит |
| Фриспины при регистрации | Не доступны |